Nylia 1/35
Generic name:norethindrone and ethinyl estradiol
Dosage form: tablets
Drug class:Contraceptives
Medically reviewed by Drugs.com. Last updated on Dec 1, 2021.
On This Page
Cigarette smoking increases the risk of serious cardiovascular events from combination oral contraceptive use. This risk increases with age, particularly in women over 35 years of age, and with the number of cigarettes smoked. For this reason, combination oral contraceptives, including NyliaTM, should not be used by women who are over 35 years of age and smoke.
Patients should be counseled that this product does not protect against HIV infection (AIDS) and other sexually transmitted diseases.
COMBINED ORAL CONTRACEPTIVES
The following product is a combined oral contraceptive containing the progestational compound norethindrone and the estrogenic compound ethinyl estradiol.
NyliaTM 1/35 Tablets:
Each peach tablet contains 1 mg of norethindrone USP and 0.035 mg of ethinyl estradiol USP. Inactive ingredients include anhydrous lactose, FD&C Yellow # 6 alumimum lake, lactose monohydrate, magnesium stearate and pregelatinized starch (maize). Each green tablet contains only inert ingredients, as follows: anhydrous lactose, croscarmellose sodium, FD&C Blue No. 2 aluminum lake, ferric oxide yellow, magnesium stearate, microcrystalline cellulose, and povidone.
The chemical name for norethindrone is 17-Hydroxy-19-nor-17α-pregn-4-en-20-yn-3-one, and for ethinyl estradiol is 19-Nor-17α-pregna-1,3,5(10)-trien-20-yne-3,17-diol. Their structural formulas are as follows: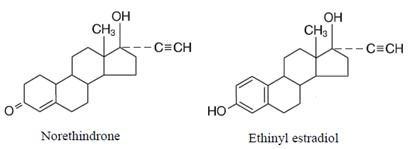
Meets USP dissolution test 3.
Nylia 1/35 - Clinical Pharmacology
Combined Oral Contraceptives
Combined oral contraceptives act by suppression of gonadotropins. Although the primary mechanism of this action is



