DexPak 13 Day TaperPak
Generic name:dexamethasone
Dosage form: tablet
Drug class:Glucocorticoids
Medically reviewed by Drugs.com. Last updated on Sep 21, 2021.
On This Page
The Dexpak Taperpak brand name has been discontinued in the U.S. If generic versions of this product have been approved by the FDA, there may be generic equivalents available.
DexPak 13 Day TaperPak Description
Dexamethasone tablets USP, 1.5mg for oral administration. Inactive ingredients are microcrystalline cellulose NF, anhydrous lactose NF, FD&C Red #40 aluminum lake, croscarmellose sodium NF, and magnesium stearate NF. The molecular weight for dexamethasone is 392.47. It is designated chemically as 9-fluoro-11β,17,21-trihydroxy-16α-methylpregna-1,4-diene-3,20-dione. The empirical formula is C22H29F05 and the structural formula is:
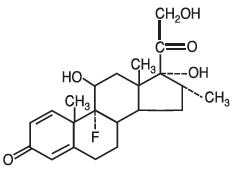
Dexamethasone, a synthetic adrenocortical steroid, is a white to practically white, odorless, crystalline powder. It is stable in air. It is practically insoluble in water.
DexPak 13 Day TaperPak - Clinical Pharmacology
Glucocorticoids, naturally occurring and synthetic, are adrenocortical steroids that are readily absorbed from the gastrointestinal tract. Glucocorticoids cause varied metabolic effects. In addition, they modify the body’s immune responses to diverse stimuli. Naturally occurring glucocorticoids (hydrocortisone and cortisone), which also have sodium-retaining properties, are used as replacement therapy in adrenocortical deficiency states. Their synthetic analogs including dexamethasone are primarily used for their anti-inflammatory effects in disorders of many organ systems. At equipotent anti-inflammatory doses, dexamethasone almost completely lacks the sodium-retaining property of hydrocortisone and closely related derivatives of hydrocortisone.



